3.6.1 Periodic motion
3.6.1.1 Circular motion
Content
- Motion in a circular path at constant speed implies there is an acceleration and requires a centripetal force.
- Magnitude of angular speed, ω = v/r
- Radian as a measure of angle.
- Direction of angular velocity will not be considered.
- Centripetal acceleration, a = v2/r = ω2r.
- The derivation of the centripetal acceleration formula will not be examined.
- Centripetal force, F = mv2/r = m ω2r.
Motion in a circular path at constant speed implies there is an acceleration and requires a centripetal force.
Acceleration is defined as the rate of change of velocity. The speed is the magnitude of the velocity. If an object is travelling at a constant speed in a circle, then its direction is constantly changing. Therefore, its velocity is constantly changing, and so must its acceleration too. For a given acceleration and mass, there must be a resultant force according to F = ma. This resultant force is referred to as the centripetal force, which acts towards the centre of the circle that the object is travelling in.
Magnitude of angular speed, ω = 2πf.
Angular speed is defined as the angular displacement per second, represented by 2π/T, and since T = 1/f, it is also equal to 2πf. It is also equal to v/r, where r is the radius of the circle being travelled in.
Radian as a measure of angle.
The radian is the standard unit of measurement of an angle.
The webpage below gives a good explanation of radians as a measure of angle.
http://www.themathpage.com/atrig/radian-measure.htm
Direction of angular velocity will not be considered.
Centripetal acceleration, a = v2/r = ω2r.
Centripetal acceleration is calculated using the formulae given above. Since ω = v/r, a can be written as ω2r instead. The centripetal acceleration can be looked at as the rate of change of the tangential velocity.
The derivation of the centripetal acceleration formula will not be examined.
Centripetal force, F = mv2/r = mω2r.
The centripetal force is calculated by using the equations above. It is the force that keeps an object moving in a uniform circular path. Since ω = v/r, then the equation can be written as mω2r. Some practice questions are given below to demonstrate use of this equation.
AQA June 2014 Unit 4 Section A Q8
Question:

Answer:
The resultant (centripetal) force is equal to mv2/r, and when the mass is as the top of the swing, both T and mg act downwards so T + mg = mv2/r, thus T = mv2/r – mg.
T must always be its lowest value at the top of the swing because the weight of the object is working with T, as opposed to working against it at the bottom, so T does not need to be as large.
The answer is 13N so B after you sub in the values to T = mv2/r – mg.

3.6.1.2 Simple harmonic motion (SHM)
Content
- Analysis of characteristics of simple harmonic motion (SHM)
- Condition for SHM: a
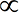 x
x - Defining equation: a = -ω2x
- x = Acosωt and
- Graphical representations linking the variations of x, v and a with time.
- Appreciation that the v-t graph is derived from the gradient of the x-t graph and that the a-t graph is derived from the gradient of the v-t graph
- Maximum speed = ωA
- Maximum acceleration = ω2A
Opportunities for skills development
- Data loggers can be used to produce s-t, v-t and a-t graphs for SHM
- Sketch relationships between x, v, a and t for simple harmonic oscillators.
Analysis of characteristics of simple harmonic motion (SHM)
For a pendulum swinging from side to side, it can be said (under certain conditions) to oscillate under simple harmonic motion (SHM). When the pendulum is at its lowest position, this can be said to be its equilibrium position, as this will be the final position of rest. An example of this is a person on a swing, the picture below demonstrates this.

Some of the key characteristics of SHM are displacement, amplitude, time period and frequency. To look as displacement, you can take the above diagram as guidance. The objects displacement will change continuously, and in one full cycle as the person on the swing starts from a position other than equilibrium will undergo the following changes.
The displacement will decrease as it moves back to its equilibrium position, and then will reverse and increase as it moves away from the equilibrium position but in the opposite direction. Then it will decrease again and then finally increase, moving towards its starting position from its equilibrium position.
Amplitude is the maximum displacement from the equilibrium position. You will get something called free vibrations if there is no frictional force and the amplitude is constant. The time period is the time taken to complete one full oscillation, denoted by the symbol T. Frequency, f, is the number of waves passing a certain point per second, and is related to time period by f=1/T. For an oscillating system you will also get an angular frequency, ω.
If you were to have two pendulums side by side with the same time period, you may be able to observe a phase difference between them. This is the difference in the fraction of the cycle that has elapsed between the two oscillating systems. This difference will be constant and is equal to the difference in time divided by the time period.
For any oscillating system, it is said that there is a restoring force that restores the object towards its equilibrium position. This resultant force must be proportional to displacement, as from equilibrium position, so that the acceleration will then be proportional to displacement.
Condition for SHM: a 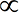 -x
-x
The condition for SHM is that the acceleration is proportional to the displacement, but acting in the opposite direction.
Defining equation: a = -ω2x
Since a ![]() –x, then a = –kx, where k is a constant. It turns out that the constant is the angular frequency squared, denoted by the ω2, where ω =2πf = v/r.
–x, then a = –kx, where k is a constant. It turns out that the constant is the angular frequency squared, denoted by the ω2, where ω =2πf = v/r.
x = Acos(ωt) and 
The equations above will find you the displacement and velocity at any point in an oscillation. x = displacement, A = amplitude, ω = angular frequency, v = velocity.
The displacement of a system oscillating with SHM can be modelled by using a sinusoidal curve, and by convention when t = 0, x = +A therefore the equation equation Acos(ωt) is used to model this situation. However if the starting point of the oscillation was chosen to be at equilibrium position moving out in a positive direction, the equation Asin(ωt) would apply.

The second equation can be derived by differentiating the first and then applying trigonometric identities. However, the derivation is not required for the course, just the application of the equation.
Graphical representations linking the variations of x, v and a with time

Appreciation that the v-t graph is derived from the gradient of the x-t graph and that the a-t graph is derived from the gradient of the v-t graph
The gradient of the displacement-time graph gives the velocity-time graph, and the gradient of the velocity-time graph derives the acceleration-time graph. If studying mathematics, knowing that differentiation can help you draw the graphs. You know that x = Acos(ωt), and so if you were to differentiate this with respect to time, you would get v = –Aωsin(ωt). The negative sign indicates that you need to draw a sine graph, but reflect it in the line y = 0. To differentiate this again with respect to time, you would get a = -Aω2cos(ωt). Thus you would draw a cosine curve reflected in the line y = 0.
Maximum speed = ωA
This equation gives you the maximum speed of an oscillating system, where ω = angular frequency and A = amplitude
Maximum acceleration = ω2A
This equation gives you the maximum acceleration of an oscillating system, where ω = angular frequency and A = amplitude
AQA Jan 2010 Unit 4 Section A Q6
Question:
Which one of the following gives the phase difference between the particle velocity and the particle displacement in simple harmonic motion?
Answer:
π/2

3.6.1.3 Simple harmonic systems
Content
- Study of mass-spring system:

- Study of simple pendulum:

- Questions may involve other harmonic oscillators (eg liquid in U-tube) but full information will be provided in questions where necessary.
- Variation of EK, EP and total energy with both displacement and time.
- Effects of damping on oscillations.
Opportunities for Skills Development
- Students should recognise the use of the small-angle approximation in the derivation of the time period for examples of approximate SHM.
Study of mass-spring system
Describing a mass-spring oscillating system can be done using the equation above, where T = Time period (s), m = mass (kg) and k = spring constant (Nm-1). The diagram below demonstrates an example of a mass-spring system.
The time period of the oscillation of a mass-spring system, unlike a pendulum is independent of g, so time periods of mass-spring systems would be the same anywhere in the universe, we think.
Furthermore, maximum tension in a spring occurs when the maximum displacement downwards is reached, and minimum displacement occurs when the spring is at maximum displacement upwards.

It can be said that the tension in the string provides the restoring force, or more specifically, the change in tension. The tension T = kΔL. When the spring is at a displacement (from equilibrium) of y, the restoring force T = -ky, where the negative sign is representative of the change in tension always attempting to bring the mass-spring system back to its equilibrium position. This then tells you that the acceleration (F/m = a), equals –kx/m.
If you follow the working out shown below, you can derive the equation a = -ω2x. This ultimately proves that the mass-spring system oscillates with simple harmonic motion.

From this equation describing the mass-spring system, you can determine how frequency of the oscillation is affected, since T = 1/f. You are able to reduce the frequency of the oscillation if you either add extra mass, or use a spring of smaller spring constant (a spring that is more easily stretched).
Study of simple pendulum: 
The equation approximating the time period of a pendulum is the same as that for the mass-spring system, but contains l instead of m, and g instead of k. The l = length of the string (m), and the g is the gravitational field strength (taken to be 9.81 unless stated otherwise). Therefore, the gravitational field strength affects the time period of a pendulum, so on the moon a pendulum would swing slower than an identical set up on earth.

Likewise, in a simple pendulum system you are able to derive the equation a = -ω2x, this working is shown below.

Questions may involve other harmonic oscillators (eg liquid in U-tube) but full information will be provided in questions where necessary.
The main oscillating systems that are dealt with are mass-spring systems and simple pendulums. However other examples are used like liquid in a U-tube, but the necessary information will be provided in questions.
https://www.youtube.com/watch?v=prZ1qbAL960 – this video provides a good explanation of SHM in a liquid U-tube.
Variation of EK, EP and total energy with both displacement and time.
As long as there is no resistance to the direction of motion of travel of say, a mass spring system, then the total energy of the system is constant, and equal to the maximum potential energy of the mass-spring system.
To consider the total energy of for example, a mass-spring system, you must first consider the variation of the kinetic energy, and potential energy of the system. The elastic potential energy EP, is given by EP = 1/2kΔL2. This is more commonly written as EP = 1/2kx2, where x is the displacement (from equilibrium).
We know that the total energy of the system is equal to the maximum potential energy of the system. This means that at maximum displacement, where x = A (amplitude), the potential energy is equal to 1/2kA2.
We also know that the total energy E = EK + EP, where EK is the kinetic energy. We can then calculate the kinetic energy as we know the total energy of the system and the potential energy. This calculation gives EK = 1/2k(A2-x2).

We also know that the maximum speed occurs when x = 0, so maximum speed occurs when v = ωA
The variation of kinetic and potential energy can be modelled on a graph of energy vs displacement. An example of this graph is shown below:

For the graphs of energy against time, to remember which line is which, you could use your knowledge that EP = 1/2kx2. If k were equal to 2, then you would have EP = x2, which could be written in the form y = x2 to make it easier to see the relationship between the parabolic curve and the equation. Hence, you get the shape of the graph shown.
On the contrary, the kinetic energy is represented by an inverted parabolic curve. This comes from the equation EK = 1/2k(A2 – x2). If you were to make k = 2, you would be left with EK = A2 – x2. For simplicity, you can look at this as y = -x2, thus an inverted parabolic curve.
The image below gives a description of the energy changes that take place during one complete oscillation of a vertical mass-spring system, as well as the velocity/acceleration at each point.
Some of the key points are that:
- At minimum displacement (x=0), the velocity is at its maximum value (ωA), thus acceleration is at its minimum value (0).
- At maximum displacement (x=A), velocity is at its minimum value (0), and so acceleration is at its maximum value (ω2A)
From these points you could draw a displacement-time, velocity-time and acceleration-time graph, as well as determine the variation of kinetic and potential energy in the system.

Effects of damping on oscillations.
Damping creates a force which acts in the opposite direction to the direction of travel of the object. This means that harmonic oscillators, in real life, will lose energy as they oscillate. This reduces the total mechanical energy of the system, thus reducing the amplitude of the oscillations. The reason for this energy loss is due to air resistance, or frictional forces. These forces dispel energy to the surrounding environment as thermal energy.
There are three types of damping: light damping, critical damping and heavy damping. The effects of each type of damping are shown in the picture below.

Source: http://www.splung.com/kinematics/images/damped_oscillations/damped_oscillations.gif
Light damping: This occurs when the time period does not change, but the amplitude decreases gradually with time. The amount that the amplitude decreases is the same fraction for each cycle.
Critical damping: This is where the damping causes the oscillating system to return back to its equilibrium position almost immediately. This type of damping is used in a car’s suspension, as you would not want the car to oscillate after it went over a bump, so critical damping returns the motion of the car back to its equilibrium state.
Over damping: This is sometimes called heavy damping. It refers to the situation in which damping is so strong that when the object is released from a non equilibrium position (ie maximum displacement), the object will return to equilibrium far slower than if it were critically damped.
3.6.1.4 Forced vibrations and resonance
Content
- Qualitative treatment of free and forced vibrations.
- Resonance and the effects of damping on the sharpness of resonance.
- Examples of these effects in mechanical systems and situations involving stationary waves.
Opportunities for Skills Development
- Investigation of the factors that determine the resonant frequency of a driven system.
Qualitative treatment of free and forced vibrations.
Free vibrations are oscillations in which the total mechanical energy of the system remains the same over time. The consequence of this is that the amplitude will remain constant throughout the oscillations. However, this idea is purely theoretical as in real life systems, energy is always dissipated to the surroundings over time, so amplitude will fall to 0. Essentially, free vibrations can be looked at as being undamped (no damping present).
In damped oscillations, clearly the amplitude will fall to 0 over time. However, in addition to the damping, if we add energy into the system to keep it oscillating, then we could say that we are providing a periodic force. A periodic force is, as it sounds, a force applied at unvarying intervals.
In forced vibrations, as the applied frequency increased from 0, the amplitude of the oscillations would increase until a maximum, and then the amplitude would decrease again. When the phase difference between the displacement and applied periodic force is p/2, the amplitude will be at its maximum. This will then increase to p as the frequency is increased.
Resonance and the effects of damping on the sharpness of resonance.
When a system oscillates with resonance, it is oscillating at maximum amplitude, as the phase difference is π/2. Essentially, the velocity and periodic force are in phase. Resonant frequency is the name given to the frequency at which maximum amplitude occurs.
The natural frequency of an oscillating system is the frequency at which the system will oscillate when not exposed to a constant or recurrent external force. The natural frequency can only be reached when little/no damping is present. This also means that for an oscillating system at resonance, the applied frequency is equal to the natural frequency.
If a system is lightly damped, then it will have a larger maximum amplitude at resonance. Also, more damping means that the resonant frequency produced will be further from the natural frequency of the system. This means that the resonance curve is sharper when damping is lighter.
The amplitude of oscillations will decrease drastically as the applied frequency becomes larger than the resonant frequency.
Examples of these effects in mechanical systems and situations involving stationary waves.
Barton’s pendulum is an example of a resonance in a mechanical system. The diagram below shows a single driver pendulum (X), and six other pendulums (ABCDEF). Clearly, X has the same length as D.

When the driver pendulum is displaced, it will cause the other pendulums to oscillate. However, pendulum D will respond more than any of the other pendulums as its length is very similar to that of D. The consequence of their similar length is that the two pendulums will have the same time period, and so their natural frequencies will be the same. This means D oscillates in resonance with X, and so has the largest amplitude. The other pendulums will respond according to their length, and how far they are from X.
Oscillations can also occur in bridges, and if they are not damped, can result in bridges oscillating at resonance if the correct periodic force is applied. This is very dangerous and can be caused by crosswinds, or people walking in step with each other.
AQA Jan 2011 Q5c
Question:
‘The length of PQ is 0.40 m. When the wire is vibrating, transverse waves are propagated along the wire at a speed of 64 ms-1. Explain why the wire is set into large amplitude vibration when the frequency of the a.c. Supply is 80 Hz.’
Answer:
- Wavelength (λ) of waves 64/80 = 0.80m
- Length of wire is λ/2 causing fundamental vibration because natural frequency of wire 64/80 = 80Hz
- Wire resonates (at frequency of ac supply), or a statement that fundamental frequency (or a natural frequency) of the wire is the same as applied frequency.
3.6.2 Thermal physics
3.6.2.1 Thermal energy transfer
Content
- Internal energy is the sum of the randomly distributed kinetic energies and potential energies of the particles in a body.
- The internal energy of a system is increased when energy is transferred to it by heating or when work is done on it (and vice versa), eg a qualitative treatment of the first law of thermodynamics.
- Appreciation that during a change of state the potential energies of the particle ensemble are changing but not the kinetic energies. Calculations involving transfer of energy.
- For a change of temperature: Q = mcΔθ where c is specific heat capacity
- Calculations including continuous flow
- For a change of state Q = ml where l is the specific latent heat.
Opportunities for Skills Development
- Investigate the factors that affect the change in temperature of a substance using an electrical method or the method of mixtures.
- Students should be able to identify random and systematic errors in the experiment and suggest ways to
- Investigate, with a data logger and temperature sensor, the change in temperature with time of a substance undergoing a phase change when energy is supplied at a constant rate.
Internal energy is the sum of the randomly distributed kinetic energies and potential energies of the particles in a body.
The internal energy of a system is the sum of the randomly distributed kinetic energies and potential energies of the particles in a body. The internal energy of a system can be thought of as the thermal energy, although some of an objects internal energy may be as a result of other factors.
The internal energy of a system is increased when energy is transferred to it by heating or when work is done on it (and vice versa), eg a qualitative treatment of the first law of thermodynamics.
There are two ways to increase the internal energy, the first of which is to do work on an object, the second of which is to transfer energy to the object by heating it. The internal energy of a system can be decreased when work is done on another object.
An example of energy transfer is in a filament lamp. Work is done by the electricity supply on the filament as electrons are pushed through. This causes the internal energy of the filament to increase, until it eventually emanates light and thermal energy to its surroundings.
Thermodynamics is a branch of physics that deals with the energy and work of a system.
The first law of thermodynamics states that the change of internal energy of an object is equal to the total energy transfer due to work done and heating.
Appreciation that during a change of state the potential energies of the particle ensemble are changing but not the kinetic energies. Calculations involving transfer of energy.
When a solid is given enough energy, the atoms will vibrate so rigorously that they may have enough energy to break free from each other. This will change the state of the solid into a liquid. The energy given to the solid raises the potential energy of the molecules as they break free from each other, although the kinetic energy does not change. The forces between molecules of a liquid are of insufficient strength to hold the molecules in fixed positions, as in a solid. On the contrary, molecules in a gas pretty much have no forces of attraction between them.
For a change of temperature: Q = mcΔθ where c is specific heat capacity
The specific heat capacity, c, of a substance is defined as the energy required to raise the temperature of 1kg of substance by 1°C/1K. Q = energy (J), m = mass (kg), q = temperature (1°C/1K) and the unit of specific heat capacity is either Jkg-1K-1 or Jkg-1°C-1, depending on the unit of temperature used. It does not matter whether Δθ is in Kelvin or Celsius because the difference between each unit on either scale is the same, ie a rise in temperature of 1K is of the same magnitude as a rise in temperature of 1°C. Although certain questions may require answers in Kelvin/Celsius.
Experimental methods to verify the specific heat capacity of a material.
- The inversion tube experiment can be used, knowing only the length of the tube, the temperature increase of the lead, and the number of inversions.
- An object ie a lead shot, is dropped from the top of the tube and falls to hit the lead shots at the bottom of the tube. The tube has a bung at the top to prevent the lead shot escaping the tube. Each time the shot hits the bottom, the tube must be flipped vertically, then once the lead shot reaches the bung it must be flipped again. This must be repeated a number of times ie 50 times, and the temperature rise noted.
- The total loss in gravitational potential energy should be equal to the gain of internal energy within the lead shots at the bottom of the tube.
- So for example, 50 inversions, the loss of gravitational potential energy is 50mgL, where L is the length of the tube.
- The gain in internal energy will be mcΔT
- So mcΔT = 50mgL, so c = 50gL/ΔT

https://revise.im/physics/unit-5/thermal-energy
These final two methods are electrical calorimeters used to determine the specific heat capacity of a solid and a liquid respectively.
- The electrical energy supplied by the heater, assuming no thermal energy is lost to the surroundings, will all be transferred to the metal block. The thermometer can be used to measure the temperature difference, so it can be said that IVt = mcΔT , so c = IVt/mΔT It is assumed that the mass of the block is known and of value m. The electrical energy supplied can be calculated using a stopwatch, ammeter and voltmeter.
- To improve the thermal conductivity of the thermometer and metal block, you can put water/oil in the hole you place the thermometer inside.

http://physicsmax.com/measure-specific-heat-capacity-metal-solid-block-method-8008
- Measurements to determine the specific capacity of a liquid can be made in a similar procedure to that above. If you take into account the energy required to heat the calorimeter, then you are left with IVt = m1c1ΔT + m2c2ΔT, where c1 is the specific heat capacity of the liquid, and all other variables are known.
- It is important to stir the water at regular intervals.

http://physicsmax.com/measure-specific-heat-capacity-metal-solid-block-method-8008
The picture below gives a comparison of the two electrical methods of determination.

http://www.schoolphysics.co.uk/age16-19/Thermal%20physics/Heat%20energy/text/Specific_heat_capacity_measurement/index.html
Calculations including continuous flow
An electric shower is an example of a continuous flow hot water system, where hot water is delivered continuously at a constant temperature and with no requirement for storage. Continuous flow only uses energy when the heat is necessitated ie when the shower is turned on.
The picture below gives a good explanation of a continuous flow heater.

http://www.schoolphysics.co.uk/age16-19/Thermal%20physics/Heat%20energy/text/Specific_heat_capacity_measurement/index.html
The energy supplied to the liquid is equal to IVt = mcDT. This process is also used in solar heating panels.
For a change of state Q = ml where l is the specific latent heat.
The equation above describes the energy changes when a solid/liquid/gas undergoes a change of state. Q is the energy required to change state, m is the mass of the substance and l is either the specific latent heat of fusion or or vaporisation (depending on which change of state is taking place).
When a solid reaches its melting point, it requires a certain amount of energy to melt it into a liquid. This energy required is called the latent heat of fusion, as a solid ‘fuses’ into a liquid when it melts. On the reverse, when a liquid cools to become a solid, latent heat is released. At the melting point, the molecules in the liquid are moving slow enough for bonds to form and keep the molecules together.
The word ‘latent’ means hidden, because when for example a solid reaches its melting point, the energy required to cause the change of state does not cause an increase in temperature of the solid.
For a liquid heated at its boiling point, the energy required to vaporise the liquid is referred to as the latent heat of vaporisation. This will occur once the molecules have been given enough kinetic energy to break free of the bonds holding them in their liquid form. On the contrary, latent heat will be released as for example, water vapour, condenses into its liquid form. As a rule of thumb, it requires more energy to vaporise a substance than melt it. Water vapour (steam) given off from a kettle is extremely hot, compared with ice that has just melted into a liquid.
There is a also a phenomenon called sublimation which occurs in some solids. In these solids, instead of changing state to a liquid, when heated they are vaporised directly instead.
By definition, ‘the specific latent heat of fusion, l, of a substance is the heat required to change a unit mass of a substance from a solid, at its melting point, into a liquid, whilst remaining at the same temperature.’
Be definition, ‘the specific latent heat of vaporisation, l, of a substance is the heat required to change a unit mass of a substance from a liquid at its boiling point, into a vapour, whilst remaining at the same temperature.’
A graph of temperature vs energy input is shown below for the changes of state from a solid to a gas.

http://www.splung.com/content/sid/6/page/latentheat
Let the substance in the graph shown be called X. At A, X is a solid and is heated towards its melting point at B. During this stage, and until C, the solid will receive latent heat of fusion, which will go towards changing the state of the solid. During this time, it is clear from the graph that the temperature does not change. Substance X is now a liquid at C, and is heated towards its boiling point at D. From D to E the liquid is provided with latent heat of vaporisation, and the liquid will change state towards becoming a gas. The temperature can be increased further towards F, but no more changes of states will occur unless the substance is contained and cooled.
3.6.2.2 Ideal gases
Content
- Gas laws as experimental relationships between p, V, T and the mass of the gas.
- Concept of absolute zero of temperature.
- Ideal gas equation: pV = nRT for n moles and pV = NkT for N
- Work done = p∆V
- Avogadro constant NA, molar gas constant R, Boltzmann constant k
- Molar mass and molecular mass.
Gas laws as experimental relationships between p, V, T and the mass of the gas.
The pressure within a gas is the force per unit area that the gas exerts on a surface. This force acts perpendicularly to the surface in which the gas is in contact with. The units of pressure, p are pascals (Pa), which can be written as Nm-2. The pressure within a gas is caused by the molecules hitting the surfaces of a container, each individual collision is insignificant, but due to the vast number of impacts occurring every second the pressure is detectable. Another thing worth noting is that the gas molecules bounce off the surfaces, or each other, without losing speed.
It can be said that the pressure of a gas depends on the volume, the temperature and the mass of the gas (in a given container). Moreover, the values of each of these properties determines the state of the gas.
The relationships between p, V, T and the mass of the gas was first established by Boyle and Charles.
Boyles Law states that, for a gas of constant mass and temperature, pV = k, where k is a constant. Any gas that obeys Boyle’s law is referred to as an ideal gas.
Charles Law states that for a given mass, at constant pressure, volume is proportional to temperature. This can be written as V/T = k2 where k2 is a constant.
The pressure law states that for a given mass at constant volume, pressure is proportional to temperature. This can be written as p/T = k3 where k3 is a constant.
Any changes that occur at a constant temperature are called isothermal changes. On the contrary, an isobaric change is one which takes place at constant pressure.
Concept of absolute zero of temperature.
Absolute zero is the lowest possible theoretical temperature. This temperature is equal to 0K on the Kelvin scale, which is equivalent to -273.15 °C. A graph of volume against temperature will yield a straight line (Charles law states they are proportional under certain circumstances). This graph relates to the temperature of ‘absolute zero’ because when volume is 0, temperature in Kelvin will be 0 ie at the origin. Although this is only followed by ideal gases.

This means that no matter how much gas is present, as long as it is an ideal gas, at absolute zero its volume will be 0.
Ideal gas equation: pV = nRT for n moles and pV = NkT for N molecules.
The three experimental laws covered previously can be combined to give the ideal gas equation pV/T = constant, which is only true for a fixed mass of an ideal gas. p is the pressure, V is the volume and T is the absolute temperature. The constant is equal to nR, where n is the number of moles and R is the molar gas constant. Thus the equation can be re-written as pV = nRT.
Something worth noting is that any ideal gases of the same volume, temperature and pressure will have the same number of moles. Rearranging the pV = nRT to pV/RT = n represents this.
For n =1, ie for 1 mol of any ideal gas in a container, pV/T = R. This value of R can be calculated to be equal to 8.31Jmol-1K-1 (given in the formula sheet). This value is called the molar gas constant, and a graph of pV vs T yields a straight line through absolute zero with a gradient = nR.
It can be said that the number of moles, n = N/NA, where N is the number of molecules and NA is Avogadro’s constant (covered in the next section). If you substitute this value of n into pV = nRT, you get pV = NRT/NA, and it can then be said that pV = NkT, because k = R/NA, where k is the Boltzmann constant, and N is the number of molecules.
Work done = p∆V
For a gas, the work done is the product of the pressure and volume during a change of volume.
Work done is defined as the product of the force acting through a distance s, where W = Fs. The units can be written in SI form as Nm.
The unit of pressure can be written as Nm-2, and the unit of volume as m3. The combined unit gives you Nm, so the unit of work.
Avogadro constant NA, molar gas constant R, Boltzmann constant k
The Avogadro constant, NA is the number of atoms in 12g of 12C equal to 6.023 x 1023. From this you can work out the mass of one atom of carbon 12 to be 12/6.023 x 1023 = 1.993 x 10-23 g. We will also use, in a later topic, something called the atomic mass unit, denoted by the symbol u. This atomic mass unit is 1/12th the mass of carbon-12. The value of u = 1.661 x 10-27 kg.
The molar gas constant was covered previously, so to recap:
For n =1, ie for 1 mol of any ideal gas in a container, pV/T = R. This value of R can be calculated to be equal to 8.31Jmol-1K-1 (given in the formula sheet). This value is called the molar gas constant, and a graph of pV vs T yields a straight line through absolute zero with a gradient = nR.
The Boltzmann constant (k = 1.38 x 10-23) relates the average kinetic energy of particles in a gas with the temperature of a gas. The Boltzmann constant k = R/NA, so the constant is a combination of two other constants.
Molar mass and molecular mass.
One mole of a substance is defined as the mass of a substance containing the same number of fundamental units as there are atoms in exactly 12g of 12C. These fundamental units could be atoms or molecule depending on the question. This means that one mole of a substance contains 6.023 x 1023 atoms/molecules. ‘Molarity’ is the number of moles in a certain amount of substance, with the unit ‘mol’.
The mass of one mol of a substance is called its molar mass, units kgmol-1. The molar mass of sulfur is roughly 0.032kgmol-1, so 0.032kg of sulfur contains 6.023 x 1023 particles.
To find the molarity of a substance, you must divide the mass of the substance (mS) by the molar mass of the substance (M). ie n = mS/M. You can find the number of molecules in a given mass by multiplying mS/M by Avogadro’s number, so N = NAmS/M.
On the contrary, molecular mass is simply the mass of a molecule. It is found by summing the constituent masses of the atoms within a molecule.
3.6.2.3 Molecular kinetic theory model
Content
- Brownian motion as evidence for existence of atoms.
- Explanation of relationships between p, V and T in terms of a simple molecular model.
- Students should understand that the gas laws are empirical in nature whereas the kinetic theory model arises from theory.
- Assumptions leading to
 including derivation of the equation and calculations.
including derivation of the equation and calculations. - A simple algebraic approach involving conservation of momentum is required.
- Appreciation that for an ideal gas internal energy is kinetic energy of the atoms.
- Use of average molecular kinetic energy =

- Appreciation of how knowledge and understanding of the behaviour of a gas has changed over time.
Brownian motion as evidence for existence of atoms.
The existence of atoms and subsequently molecules was first established in 1827, by Robert Brown. His observation of pollen grains in water showed the irregular random motion of particles in a fluid to be as a result of continuous bombardment of molecules in the surrounding medium.
Explanation of relationships between p, V and T in terms of a simple molecular model.
The pressure of a gas at constant temperature can be increased by decreasing the volume in which the gas is contained. This is because the molecules within the container need to travel a smaller distance between impacts on the surface of the container, so there are more impacts per unit time, and so an increased pressure. This explains Boyle’s Law.
Increasing the temperature of a gas increases the average speed, so the impacts within the container are more frequent and of larger significance. This increases pressure and relates to the pressure law.
Students should understand that the gas laws are empirical in nature whereas the kinetic theory model arises from theory.
The gas laws are empirical in a nature; ie they have been found experimentally. Whereas, the kinetic theory model arises from mathematics and theories.
Assumptions leading to pV =  including derivation of the equation and calculations.
including derivation of the equation and calculations.
In deriving this equation shown above, you must first make assumptions about the molecules within a gas. These assumptions are as follows:
- You can treat molecules as point objects, with a negligible volume relative to the volume of the gas
- There are no attractive forces between the molecules within the gas. If there were attractive forces the impact forces on the surfaces of the container would be reduced
- Their motion is continuous and completely unpredictable
- All collisions, either between molecules or molecules and the container surface, are elastic. This means there is no loss in kinetic energy after collisions
- Finally, the duration of contact between molecules and the container surface is negligible compared to the time between impacts.
A simple derivation of the equation pV =![]() is given below. In this derivation, the velocity in the x direction is represented by vX, in the y direction by vY and z direction by vZ.
is given below. In this derivation, the velocity in the x direction is represented by vX, in the y direction by vY and z direction by vZ.
Also, Pythagoras’ theorem, as noted in the derivation, can be applied to three dimensions. Finding the resultant velocity in 2 dimensions can be done by doing v2 = (vx)2 + (vY)2, and this can be extended in three dimensions so v2 = (vx)2 + (vY)2 + (vZ)2 as the individual velocities are all perpendicular to each other. Other than that the derivation should be trivial.


A simple algebraic approach involving conservation of momentum is required.
In the derivation above, momentum is treated algebraically. This is because the actual speed and mass of the particle is unknown, thus are denoted by m and v respectively. This means that change in momentum = -mv – (mv) = -2mv.
Appreciation that for an ideal gas internal energy is kinetic energy of the atoms.
For an ideal gas, the internal energy should be due only to the kinetic energy of the atoms. As the temperature of a gas increases, the average kinetic energy of the molecules within a container will increase.
Use of average molecular kinetic energy = 
The average molecular kinetic energy is given by which is also equal to , equal also to . The derivation of these equations is not required, only their application. Each symbol has the same associated unit and name as mentioned already.
Leave a comment